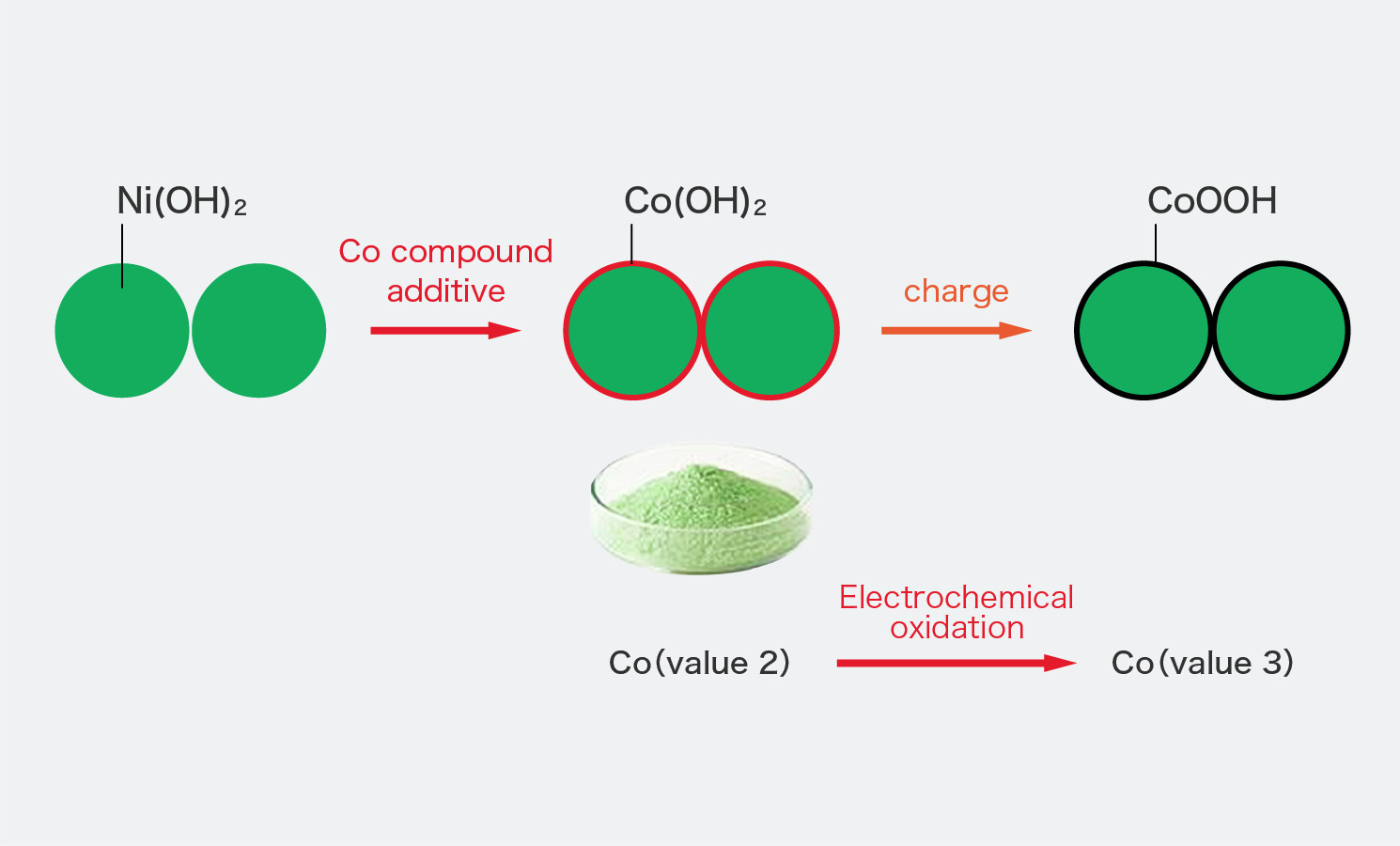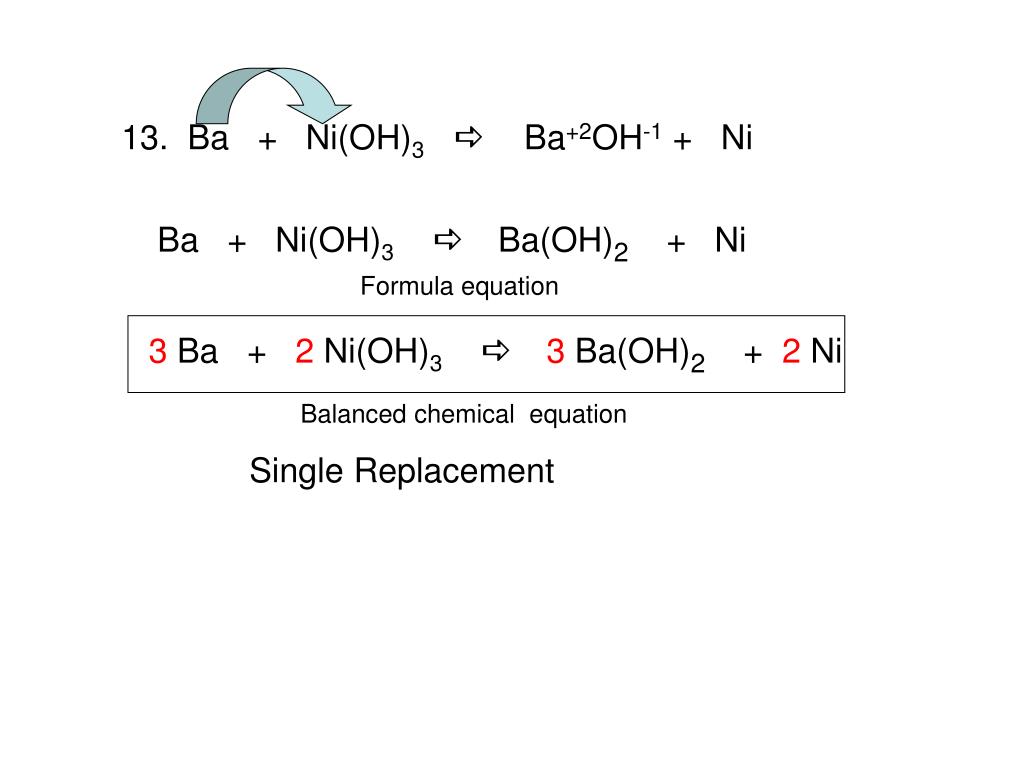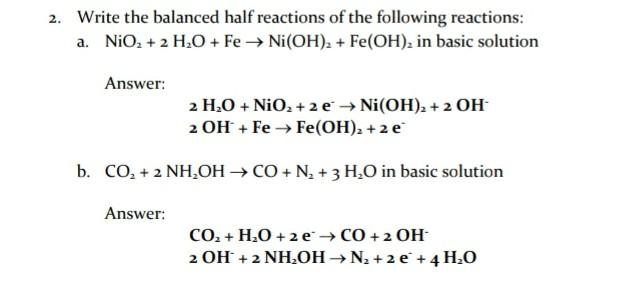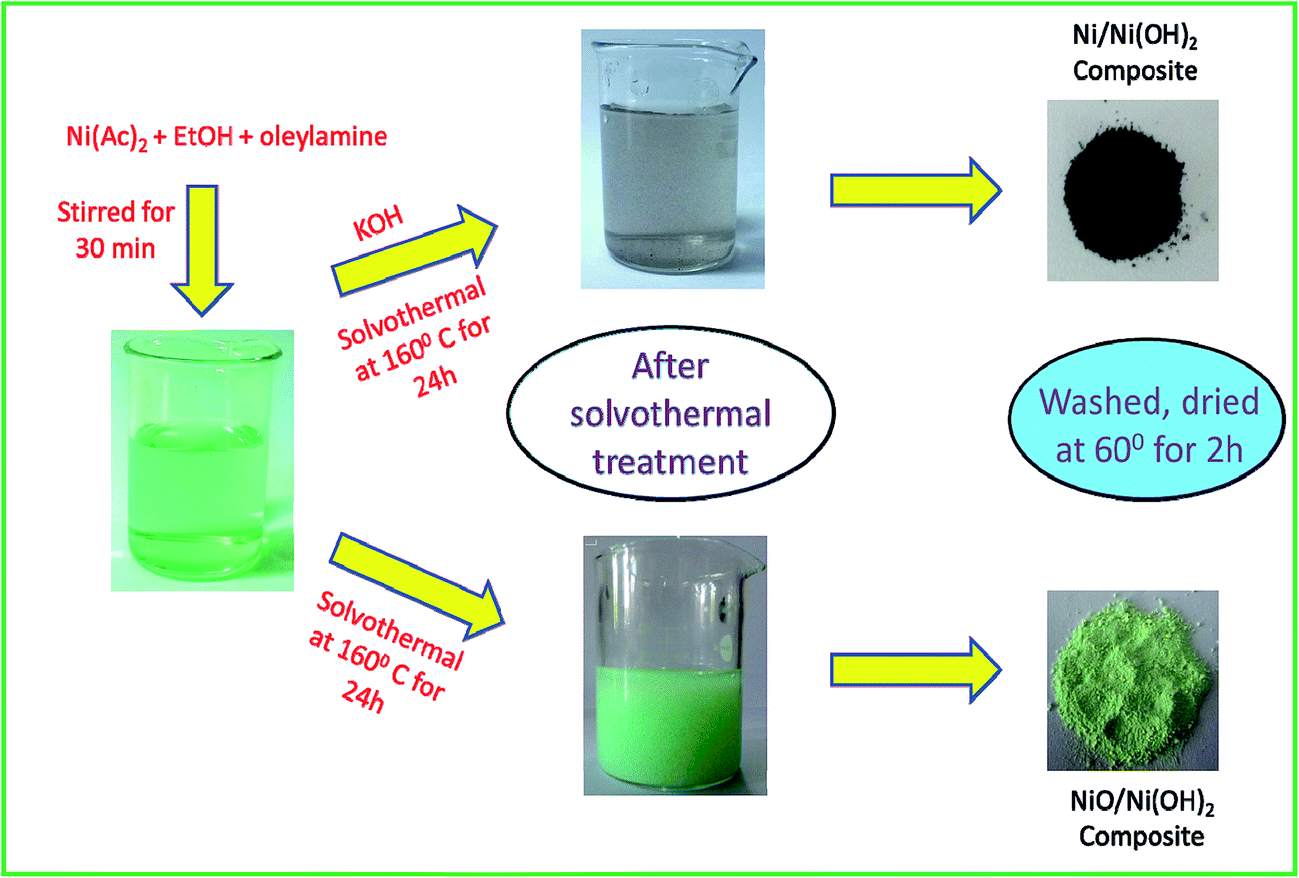
One step synthesis of Ni/Ni(OH) 2 nano sheets (NSs) and their application in asymmetric supercapacitors - RSC Advances (RSC Publishing) DOI:10.1039/C6RA26584G
![The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/5a5d2b1b-25f4-4e6d-a83c-c65beeb759da/chem202203128-fig-0003-m.jpg)
The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library

XRD patterns of (a) In(OH)3, (b) Ni(OH)2, (c) Ni(OH)2/In(OH)3, and (d)... | Download Scientific Diagram

Facile preparation of three-dimensional Ni(OH)2/Ni foam anode with low cost and its application in a direct urea fuel cell - New Journal of Chemistry (RSC Publishing)
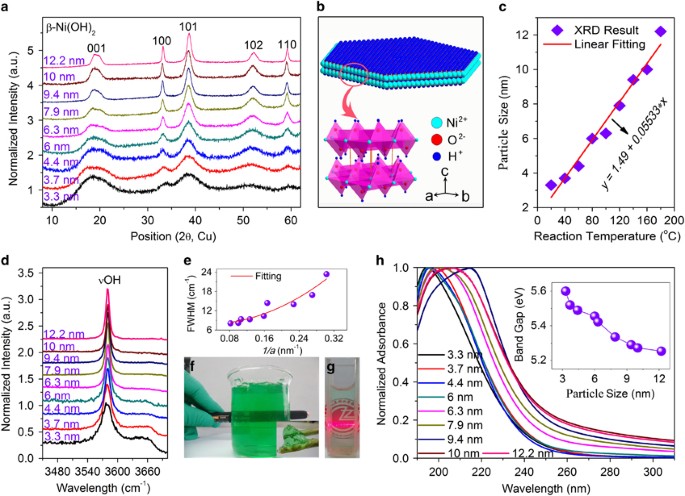
Ultra-small, size-controlled Ni(OH)2 nanoparticles: elucidating the relationship between particle size and electrochemical performance for advanced energy storage devices | NPG Asia Materials
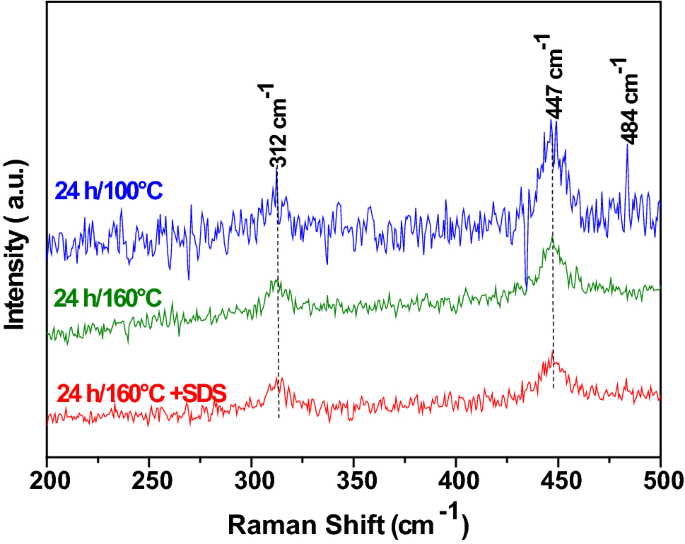
Electrochemical measurements of synthesized nanostructured β-Ni(OH)2 using hydrothermal process and activated carbon based nanoelectroactive materials | SN Applied Sciences

Fabrication of Three-Dimensional Porous NiO/Amorphous Ni(OH)2 Composites for Supercapacitors | Energy & Fuels

K of a salt Ni(OH), is 2 x10-15 then molar solubility of Ni(OH), in 0.01M NaOH is :- (1) 2 x 10-15 M (2) 21/3 x 10-5 M (3) 2 x 10-11 M (4) 107 M
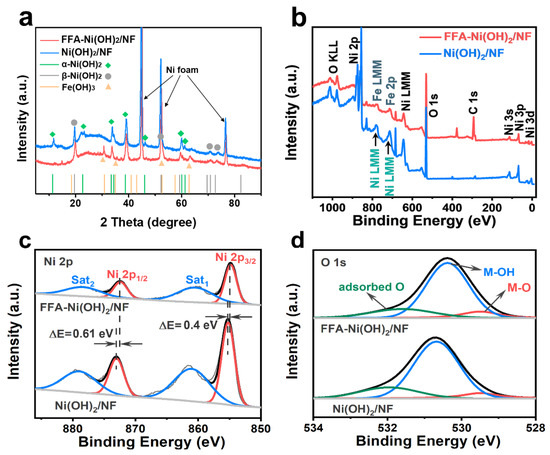
Crystals | Free Full-Text | Ferrocene Formic Acid Surface Modified Ni(OH)2 for Highly Efficient Alkaline Oxygen Evolution

Chemistry lovers - #Metal hydroxides are hydroxides of metals. Metal hydroxides are also known as strong bases. Many common metal hydroxides are made up from hydroxide ions and the ion of the

Nickel hydroxides and related materials: a review of their structures, synthesis and properties | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences

Direct preparation of Al-substituted α-Ni(OH)2 from Al-containing salt solution by immersing method - ScienceDirect